Technology Type
- Type
- Chlor-Alkali Process
- Process
- Decompositions reactions
- Abbreviation
-
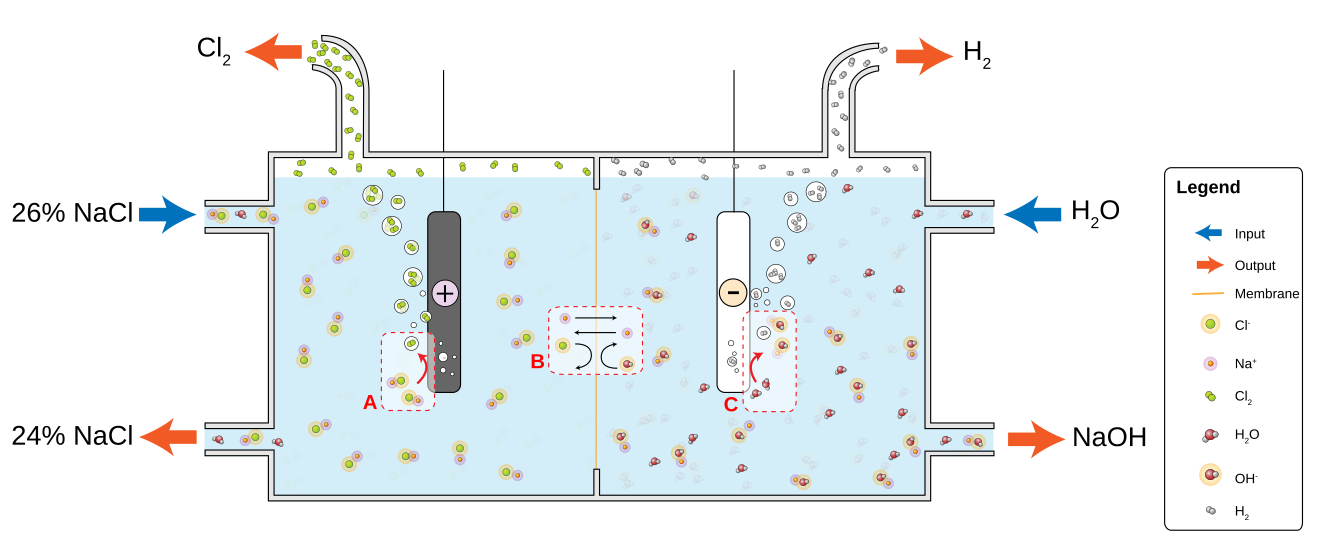
The Chlor-Alkali Electrolysis Process is used in the Manufacture of Chlorine, Hydrogen, and Sodium Hydroxide (Caustic) solution. Of these 3, the primary Product is Chlorine. There are 3 types of Electrolytic Processes used in the Production of Chlorine:
- the Diaphragm Cell Process,
- the Mercury Cell Process, and
- the Membrane Cell Process.
In each Process, a Salt Solution is electrolyzed by the action of direct electric current that converts Chloride Ions to Elemental Chlorine. The overall Process Reaction is:
2 NaCl + 2 H2O → Cl2 + H2 + 2 NaOH
In all 3 methods, the Chlorine (Cl2) is produced at the Positive Electrode (Anode) and the Caustic Soda (NaOH) and Hydrogen (H2) are produced, directly or indirectly, at the Negative Electrode (Cathode). The 3 Processes differ in the method by which the Anode Products are kept separate from the Cathode Products.
- Link
System Info
- Updated by
-
 Kokel, Nicolas
Kokel, Nicolas - Updated
- 1/10/2023 7:50 AM
- Added
- 4/2/2022 2:12 PM

No Services yet available.
Enquire in Solutions how we can help you.
Technology Type Communicator
| Title | Date |
|---|
Image
Technologies
| Technology | Owner | |
|---|---|---|

|
Dow Inc. |