Natural rubber derived from the latex of Hevea brasiliensis (commonly known as the Pará rubber tree or rubber tree) is the world's most commercially significant elastomeric biopolymer, accounting for approximately 40–45% of total global rubber consumption. It is sourced by tapping the bark of the tree to collect the milky-white latex sap, which is a colloidal aqueous suspension containing roughly 30–40% dry rubber content, along with proteins, lipids, carbohydrates, and inorganic salts.
Chemical Composition
At the molecular level, Hevea natural rubber is composed almost entirely of cis-1,4-polyisoprene — a stereoregular polymer in which isoprene monomer units (C₅H₈) are linked exclusively in the cis configuration across the double bond. This high degree of stereochemical regularity, with cis content exceeding 99.9%, is what distinguishes it from synthetic polyisoprene and underpins its extraordinary elastic properties. The molecular weight of commercial Hevea rubber typically ranges from 200,000 to over 8,000,000 g/mol depending on the tree clone and processing method.
Physical & Mechanical Properties
Hevea rubber exhibits a unique combination of properties that few synthetic alternatives can fully replicate:
- High elasticity — capable of stretching to 5–8 times its original length and recovering almost completely
- Tensile strength — up to 24–30 MPa in vulcanized form
- Low heat build-up — excellent hysteresis characteristics, critical for dynamic applications like tires
- Resilience — superior rebound and fatigue resistance under repeated stress
- Tack — natural self-adhesion, important in tire building and adhesive applications
- Low temperature flexibility — retains elasticity down to approximately −50°C
Processing
Raw latex is processed into several commercial forms. It is either preserved as liquid concentrated latex (for dipping, foaming, or adhesive applications) or coagulated and dried into solid forms such as Ribbed Smoked Sheet (RSS), Technically Specified Rubber (TSR), or crepe rubber. Vulcanization — the cross-linking of polymer chains with sulfur under heat — is the critical step that converts raw rubber into a durable, temperature-stable, and chemically resistant product.
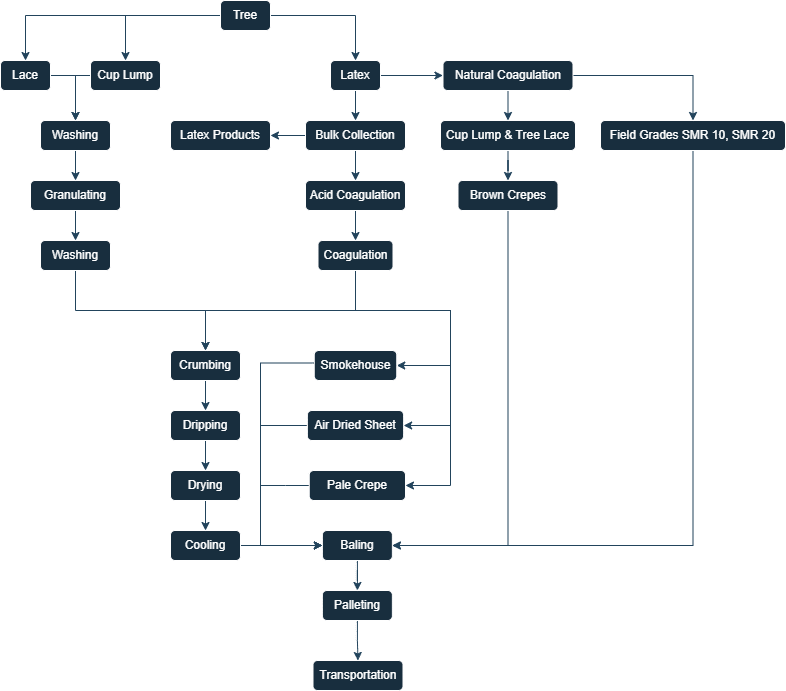
Figure 1 — Flow chart of natural rubber processing [5]
Key Applications
Hevea-derived natural rubber is indispensable in high-performance applications where synthetic rubbers fall short:
- Tires — particularly aircraft, truck, and off-road tires requiring high load and heat resistance
- Medical devices — surgical gloves, catheters, and condoms (though latex allergy is a concern)
- Industrial engineering — conveyor belts, engine mounts, vibration dampers, and seals
- Adhesives and coatings — contact cements and pressure-sensitive adhesives
- Foam products — mattresses and cushioning via latex foaming processes
Limitations
Despite its outstanding properties, Hevea rubber has some notable weaknesses: poor resistance to oils, hydrocarbon solvents, ozone, and UV radiation, as well as susceptibility to oxidative degradation at elevated temperatures. Additionally, its proteins can trigger Type I latex hypersensitivity (latex allergy) in sensitized individuals, which has driven demand for alternatives such as guayule-derived rubber in medical applications.
References
- ScienceDirect. Hevea Brasiliensis (Accessed Mar 4, 2026)
- Britannica. polyisoprene (Page version: Dec 27, 2025)
- Gamelsky R., Heveaplanet.com (Jun 13, 2019). Why Natural Rubber?
- Kułakowski B., INPART24 (Mar 9, 2024). Natural rubber - NR Rubber - definition, types, properties, applications
- Rheometer Spares (Nov 17, 2023). From Tree to Tyre: The Complete Guide to Natural Rubber
- Wikipedia. Natural rubber (Page version: Feb 22, 2026)