Sodium hexafluoroaluminate (Cryolite) is produced by several different routes
- Technology Type
- Sodium Hexafluoroaluminate Synthesis
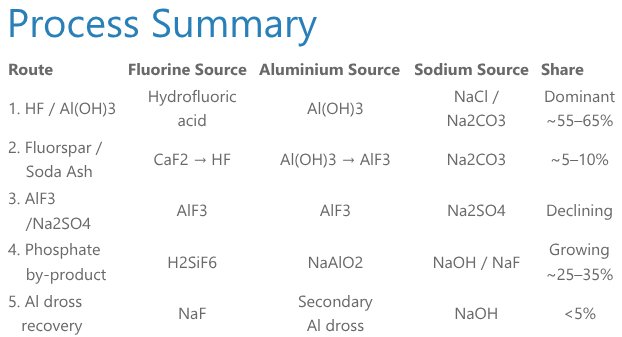
Five differet routes to produce cryolite
The synthesis of sodium hexafluoroaluminate (Na3AlF6), the IUPAC name for synthetic cryolite, is an inorganic double-replacement (metathesis) reaction in which fluoride ions progressively substitute hydroxide or oxide ligands around aluminium, while sodium cations combine with the resulting [AlF6]3- complex to precipitate the final crystalline product. All industrial routes share the same three essential inputs: a fluorine source (hydrofluoric acid or fluorosilicic acid), an aluminium source (aluminium hydroxide, sodium aluminate, or aluminium fluoride), and a sodium source (soda ash, sodium hydroxide, or sodium chloride).
Read the detailed technology profile here.
#syntheticcryolite #cryolitesynthesis #sodiumhexafluoroaluminate