Total natural CaCO₃ production (as limestone rock) exceeds 4–5 billion metric tonnes per year
- Product
- Ground Calcium Carbonate
- Message Category
-
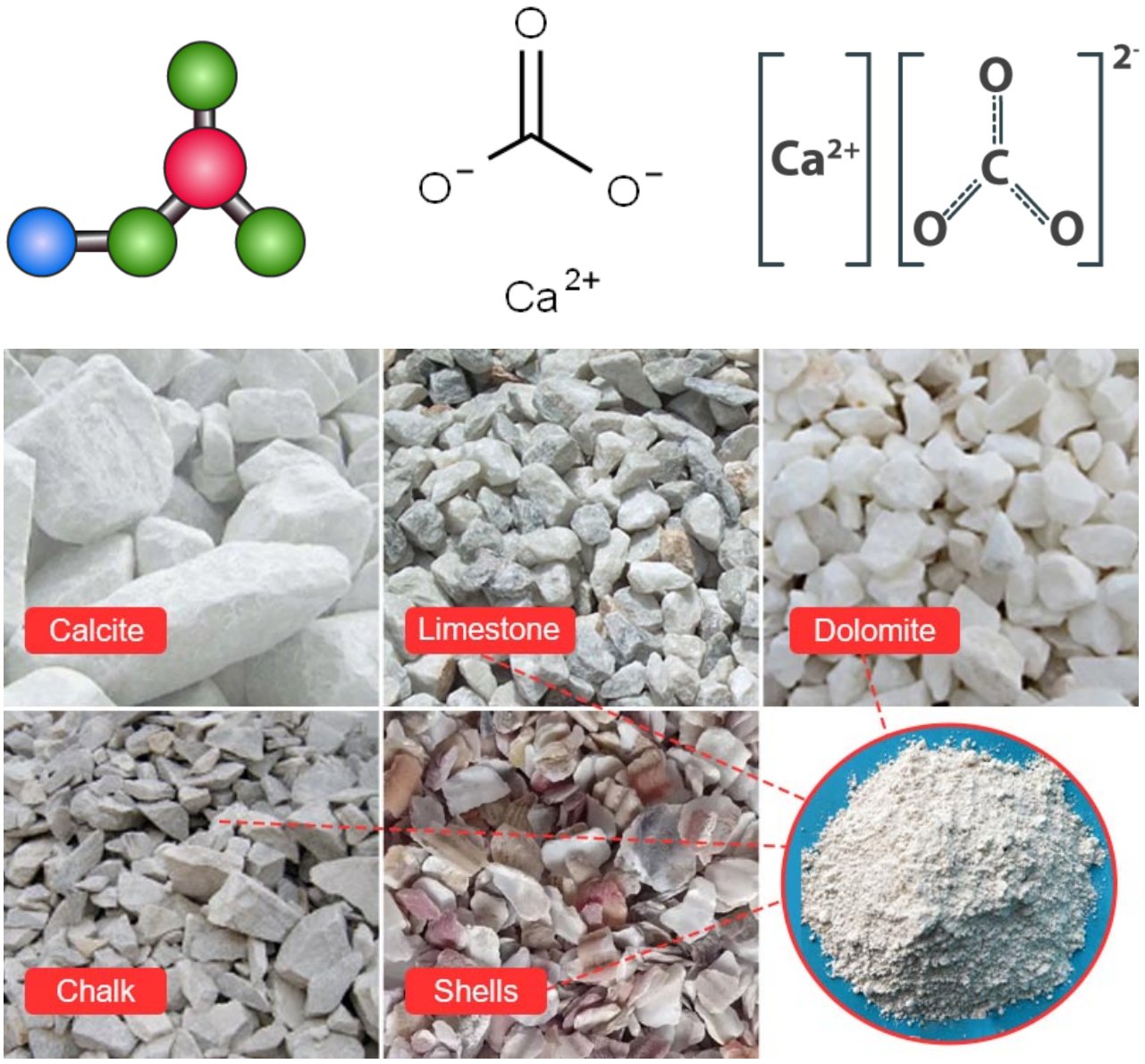
Top: molecular structure of calcium carbonate (various sources) | Bottom: raw material for ground calcium carbonate powder production (source: DASWELL)
Calcium carbonate (CaCO₃) is an inorganic chemical compound consisting of one calcium atom, one carbon atom, and three oxygen atoms, forming one of the most abundant and widely distributed substances in nature. It is the principal constituent of limestone, marble, chalk, coral, eggshells, and the shells of most marine organisms. It occurs naturally in three distinct mineral polymorphs — calcite, aragonite, and vaterite — of which calcite is the thermodynamically most stable at ambient conditions.
Read the detailed product profile here.










