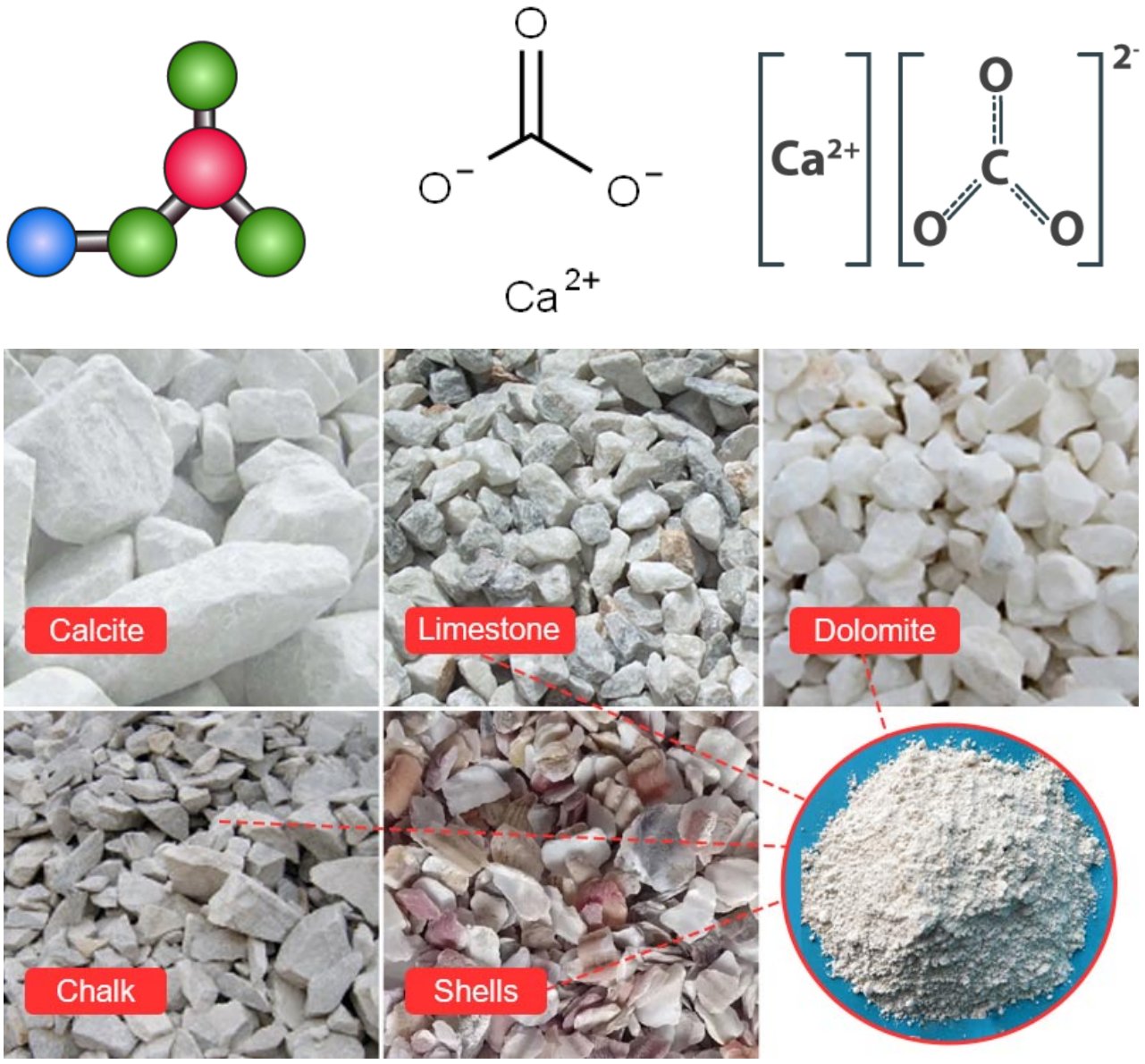
Calcium carbonate (CaCO₃) is an inorganic chemical compound consisting of one calcium atom, one carbon atom, and three oxygen atoms, forming one of the most abundant and widely distributed substances in nature. It is the principal constituent of limestone, marble, chalk, coral, eggshells, and the shells of most marine organisms. It occurs naturally in three distinct mineral polymorphs — calcite, aragonite, and vaterite — of which calcite is the thermodynamically most stable at ambient conditions.
Identifiers
| Field |
Value |
| CAS Number |
471-34-1 (pure CaCO₃);
1317-65-3 (limestone grade) |
| EC Number |
207-439-9 |
| IUPAC Name |
Calcium carbonate |
| PubChem CID |
10112 |
| Molecular Formula |
CaCO₃ |
| Molecular Weight |
100.09 g/mol |
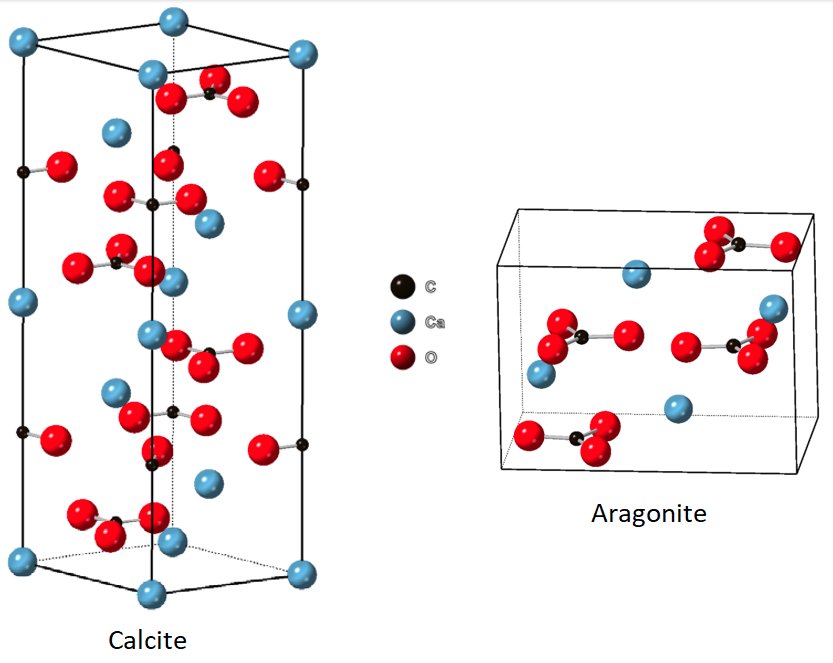
Crystal Structure & Polymorphs
Calcium carbonate crystallizes in three anhydrous polymorphs:
- Calcite — hexagonal (trigonal) system; the stable form at room temperature; density 2.71 g/cm³; space group D⁶₃d with lattice constant a = 0.636 nm
- Aragonite — orthorhombic system; slightly denser at 2.83 g/cm³; metastable at room temperature; forms at temperatures above 85°C or under high pressure
- Vaterite — hexagonal; the least stable polymorph; rare in nature; forms by precipitation at ~60°C
In calcite, each Ca²⁺ ion is coordinated by six oxygen atoms; in aragonite by nine, reflecting the different packing geometries:
Physical & Chemical Properties
| Property |
Calcite |
Aragonite |
| Appearance |
White powder /
colorless crystals |
White orthorhombic
crystals |
| Density |
2.71 g/cm³ |
2.83 g/cm³ |
| Melting / Decomposition |
Decomposes at 825°C |
Melts at 1,339°C (102.5 atm) |
| Solubility in water |
~15 mg/L at 25°C |
Slightly higher |
| Ksp |
4.8 × 10⁻⁹ |
6.0 × 10⁻⁹ |
| Odor |
Odorless |
Odorless |
| Taste |
Chalky |
Chalky |
Calcium carbonate is insoluble in pure water but dissolves readily in dilute acids and in water containing dissolved CO₂.
Key Chemical Reactions
1. Thermal decomposition (calcination) — the basis of lime and cement manufacturing:
Calcium carbonate is heated in a kiln to above 825°C, breaking its crystal lattice and driving off carbon dioxide gas to yield calcium oxide (quicklime) — the foundation of the entire lime and cement industry.
CaCO3 → CaO + CO2 (@ 825°C)
2. Reaction with acids — produces CO₂ gas (effervescence):
Two moles of hydrochloric acid attack the carbonate ion, releasing carbon dioxide as a visible gas, forming soluble calcium chloride and water — the classic "fizzing" test used to identify carbonate minerals in the field.
CaCO3 + 2 HCl → CaCl2 + H2O + CO2↑
3. Reaction with water + CO₂ — karst dissolution and cave formation:
Rainwater enriched with dissolved carbon dioxide forms weak carbonic acid, which slowly converts insoluble calcium carbonate into soluble calcium bicarbonate — the mechanism responsible for the dissolution of limestone to form caves, sinkholes, and karst landscapes.
CaCO3 + H2O + CO2 → Ca(HCO3)2
4. Reaction with sulfuric acid:
Sulfuric acid reacts with calcium carbonate to form calcium sulfate (gypsum), water, and carbon dioxide — a reaction exploited industrially in flue gas desulfurization (FGD) systems to capture SO₂ emissions, and also the cause of acid rain damage to limestone buildings and monuments.
CaCO3 + H2SO4 → CaSO4 + H2O +CO2
Commercial Forms & Grades
Two primary commercial grades are produced:
- Ground Calcium Carbonate (GCC) — mechanically ground from natural limestone, marble, or chalk; particle size ranges from 1–100 µm; used as a filler and coating pigment
- Precipitated Calcium Carbonate (PCC) — synthetically produced by carbonation of lime slurry; higher purity, controlled particle morphology; used in pharmaceuticals, food, and high-performance coatings
Industrial & Commercial Applications
Calcium carbonate is one of the most versatile industrial minerals in use, spanning multiple sectors:
Construction & Cement
- Primary raw material for Portland cement clinker production
- Filler in concrete, asphalt, and mortars
Iron & Steel
- Flux in blast furnaces and basic oxygen steelmaking to remove silica impurities as slag
Paper & Printing
Plastics & Rubber
- Filler to improve stiffness, impact resistance, and reduce cost in PVC, polyolefins, and rubber compounds
Paints & Coatings
- Extender pigment providing opacity, gloss control, and surface smoothness
Agriculture
- Agricultural limestone to neutralize soil acidity and supply calcium to crops
Environmental
Pharmaceuticals & Food
- Antacid tablets (directly neutralizes stomach acid: CaCO₃ + 2 HCl → CaCl₂ + H₂O + CO₂)
- Calcium supplement in dietary products and infant formula
- Food additive (E170) as a color, firming agent, and anti-caking agent
- Ingredient in toothpaste as a mild abrasive and whitening agent
Global Market & Production
Calcium carbonate is one of the highest-volume commodity chemicals globally. Total natural CaCO₃ production (as limestone rock) exceeds 4–5 billion metric tonnes per year. The specialty GCC/PCC market alone was valued at over USD 13.6 billion in 2024, with volume around 112 million metric tonnes for processed grades. China dominates global production, followed by the United States, India, and European producers.
References
- ALLEN Career Institute (Jul 17, 2025). Calcium carbonate
- Britannica (Feb 19, 2026). calcium carbonate
- BYJU'S (Sep 7, 2017. What is Calcium Carbonate?
- ChemicalBook. Calcium carbonate (Accessed: Mar 8, 2026)
- Covia Holdings LLC. Calcium carbonate (Accessed: Mar 8, 2026)
- National Center for Biotechnology Information (PubChem). Calcium Carbonate (Accessed: Mar 8, 2026)
- SoftSchools. Calcium carbonate Formula - Calcium carbonate Uses, Properties, Structure and Formula (Accessed: Mar 8, 2026)
- Wikipedia. Calcium carbonate (Page version: Feb 20, 2026)
- DASWELL (Jul 19, 2022). Understanding Heavy Calcium Carbonate from Six Perspectives