Introduction
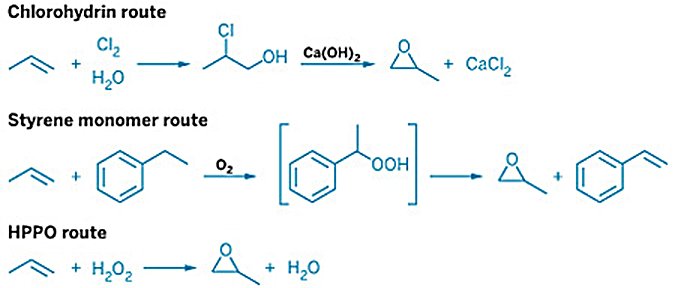
Propylene oxide (PO) is one of the chemical industry's most important intermediates, produced at over 10 million tonnes per year globally and consumed primarily as the precursor to polyurethane polyols, propylene glycol, and glycol ethers. The historical chlorohydrin process, introduced in the 1950s, was the first industrial PO-only route but generated large salt effluents and required integrated chlorine infrastructure.
To address these limitations, epoxidation routes — in which propylene is epoxidized using a preformed peroxide oxidant — were developed as cleaner, chlorine-free alternatives from the 1960s onward. These routes are the dominant commercial pathway for PO production worldwide and have displaced the chlorohydrin process for all new capacity since the early 1970s. They can be grouped into two families:
-
Organic hydroperoxide routes: propylene is epoxidized by an organic hydroperoxide derived from a hydrocarbon co-feed — ethylbenzene (SMPO), isobutane (TBA), or cumene (POC). The first two co-produce organic by-products; the cumene route recycles its carrier and produces PO as the sole product.
-
Hydrogen peroxide route (HPPO): propylene is epoxidized with hydrogen peroxide (H2O2) over a TS-1 zeolite catalyst in methanol solvent; water is the only by-product.
Principle
In all peroxidation routes, the reaction involves transfer of an electrophilic oxygen atom from the peroxide oxidant to the electron-rich propylene double bond, forming the three-membered epoxide ring of propylene oxide. The general stoichiometry is:
C3H6 + [O] → C3H6O (PO)
where RO2H is an organic hydroperoxide (ROH = co-product alcohol) or H2O2 (ROH = H2O). The peroxide is fully consumed; the oxygen-depleted carrier exits as a co-product, is recycled, or exits as water.
Process Routes
Four distinct peroxide carriers are used in commercial peroxidation processes:
| Route |
Peroxide carrier |
Co-product |
First commercial |
Licensor(s) |
| SMPO |
Ethylbenzene hydroperoxide (EBHP) |
Styrene monomer, 2.2–2.5 t/t PO |
1969 |
LyondellBasell, Shell |
| PO/TBA |
tert-Butyl hydroperoxide (TBHP) |
tert-Butanol/MTBE, 2.4–2.8 t/t PO |
1973 |
LyondellBasell, Huntsman |
| POC (Cumene) |
Cumene hydroperoxide (CMHP) |
None (cumene recycled) |
2003 |
Sumitomo Chemical |
| HPPO |
Hydrogen peroxide (H2O2) |
Water, ~0.3 t/t PO |
2008 |
BASF–Dow; Evonik–thyssenkrupp Uhde |
Reaction Mechanism
All four routes share a common catalytic mechanism: a metal–peroxo or metal–hydroperoxo species activates the peroxide oxidant and transfers the distal oxygen to the propylene double bond. The catalyst and reaction conditions differ by route:
-
SMPO and TBA routes: epoxidation catalyzed by molybdenum homogeneous compounds (Halcon-type, Mo–OOR intermediate) or titanium/silica heterogeneous catalysts (Shell-type, Ti–OOR intermediate); 90–130°C, 1.5–6 MPaG; the alcohol ROH exits as co-product.
-
POC (Cumene) route: epoxidation over Sumitomo's proprietary mesoporous Ti/SiO2 catalyst (pore diameter 1–20 nm); the large CMHP molecule cannot enter TS-1's ~5–6 Å micropores, necessitating the wider mesoporous support; CMA co-product is hydrogenated back to cumene and recycled; mild temperature, moderate pressure.
-
HPPO route: epoxidation over TS-1 (Titanium Silicalite-1) zeolite in methanol solvent; active site is a hydroperoxotitanium intermediate (Ti–OOH) at tetrahedral Ti4? framework sites; water is the only by-product; <90°C, ~30 bar.
Three competing reactions reduce PO yield in all liquid-phase peroxidation routes and must be minimized through temperature control, solvent management, and propylene excess:
-
PO + H2O → propylene glycol (1,2-propanediol) [Reaction constant: k2]
-
PO + ROH → glycol ethers or methoxypropanols [Reaction constant: k3]
-
Peroxide decomposition → unproductive oxidant loss
Individual Process Descriptions
1. SMPO Process (PO/Styrene Monomer Method)
Developed by Halcon Corp. and ARCO in the late 1960s, the SMPO process uses ethylbenzene as feedstock to co-produce PO and styrene monomer. It is currently licensed primarily by LyondellBasell and Shell.
Process steps:
-
Ethylbenzene oxidation: ethylbenzene is oxidized with air in liquid phase at 140–165°C, atmospheric pressure, without catalyst, at ~10–12% conversion per pass, to form ethylbenzene hydroperoxide (EBHP)
-
Epoxidation: EBHP reacts with propylene at 90–130°C, 1.5–6 MPaG over a molybdenum homogeneous or Ti/SiO2 heterogeneous catalyst, producing PO and α-methylbenzyl alcohol; EBHP conversion ≥95%, PO selectivity ~90%
-
Dehydration: α-methylbenzyl alcohol is dehydrated over an acid catalyst to form styrene monomer

Co-product ratio: ~2.2–2.5 t styrene per tonne PO
Economic note: SMPO economics are structurally coupled to the styrene market. Declining European competitiveness — driven by Asian styrene imports and high energy costs — led to the permanent closure of the LyondellBasell/Covestro Maasvlakte SMPO unit in March 2025.
2. PO/TBA Process (Propylene Oxide / Tert-Butanol Method)
The TBA process uses isobutane as feedstock to co-produce PO and tert-butanol (TBA), further processed to MTBE or isobutylene. It was developed by Halcon/ARCO and is operated primarily by LyondellBasell and Huntsman.
Process steps:
-
Isobutane oxidation: isobutane is oxidized with air in liquid phase at 120–140°C, 3–4 MPaG to form tert-butyl hydroperoxide (TBHP); conversion 35–50%, TBHP selectivity 50–60%
-
Epoxidation: TBHP reacts with propylene at 90–130°C, 1.5–6 MPaG over a molybdenum compound catalyst, producing PO and TBA; TBHP conversion ≥95%, PO selectivity ~90%
-
TBA utilization: TBA is dehydrated to isobutylene, which is reacted with methanol to produce MTBE, or sold directly as TBA

Co-product ratio: ~2.4–2.8 t TBA or MTBE equivalent per tonne PO
Economic note: the TBA process faces structural pressure from declining MTBE demand in markets where fuel oxygenate regulations have become increasingly restrictive.
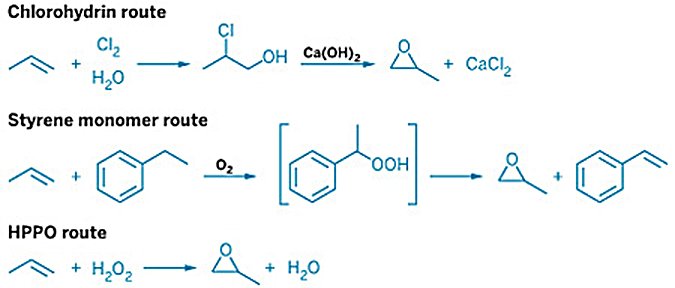
3. POC Process (Propylene Oxide via Cumene — Cumene Method)
Developed and commercialized by Sumitomo Chemical from 2003, the POC process uses cumene as a recyclable oxygen carrier. It is the first organic hydroperoxide PO route to achieve a single-product profile — PO only, with no net co-product.
Process steps:
-
Cumene oxidation: cumene is oxidized with air at 90–130°C to form cumene hydroperoxide (CMHP); selectivity to valuable oxygenated products ≥95%
-
Epoxidation: CMHP reacts with propylene over Sumitomo's proprietary mesoporous Ti/SiO2 catalyst (pore size 1–20 nm), producing PO and α,α-dimethylbenzyl alcohol (CMA); PO selectivity ≥95%
-
CMA hydrogenation: CMA is hydrogenated back to cumene in a fixed-bed reactor (conversion >99%); cumene is recycled to the oxidation step; water is the only net by-product
Co-product ratio: none; cumene fully recycled
Commercial deployments: Chiba Japan (150,000 t/y, 2003; expanded to 200,000 t/y 2005), Rabigh Saudi Arabia (200,000 t/y, 2009), Ulsan South Korea (300,000 t/y, 2018), Map Ta Phut Thailand (~200,000 t/y, ~2020)
4. HPPO Process (Hydrogen Peroxide to Propylene Oxide)
The HPPO process produces PO by direct liquid-phase epoxidation of propylene with hydrogen peroxide over a TS-1 zeolite catalyst in methanol solvent. H2O2 is produced separately via the anthraquinone oxidation (AO) process (Riedl-Pfleiderer process) in a co-located plant. Water is the only by-product. Two commercially independent HPPO technologies are available for licensing: the BASF–Dow HPPO process and the Evonik–Uhde HPPO process.
Main reaction:
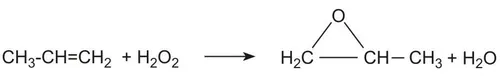
Process conditions: <90°C, ~30 bar, methanol solvent; overall H2O2 conversion >99%, PO selectivity 95–97%, PO yield 93–95% (based on H2O2); final PO purity ≥99.8 wt%
Anthraquinone process for H2O2 production (cyclic, co-located):
-
Hydrogenation: 2-alkylanthraquinone is hydrogenated over Pd catalyst at 40–50°C, 2–4 bar H2 to anthrahydroquinone
-
Oxidation: anthrahydroquinone is oxidized with air at 30–60°C, regenerating anthraquinone and producing H2O2
-
Extraction: H2O2 is extracted with water, yielding 30–70 wt% aqueous solution for direct feed to the HPPO reactor
Co-product ratio: ~0.3 t water per tonne PO (stoichiometric only)
Technology developers and licensors:
-
BASF + Dow, with Solvay as co-located HP partner — three world-scale plants in operation since 2008 at Antwerp (Belgium), Map Ta Phut (Thailand), and Jubail II (Saudi Arabia); total licensed capacity ~1,080,000 t/y.
-
Evonik Industries + thyssenkrupp Uhde, with Evonik as co-located HP partner — four plants in operation or under construction since 2008 in South Korea, China (×2), and Hungary; total licensed capacity ~930,000 t/y.
-
Sinopec — domestic HPPO technology; 100,000 t/y unit commissioned 2014
Status and Outlook
The SMPO and TBA routes remain the largest installed base globally but are in structural decline in high-cost regions such as Western Europe, driven by co-product market volatility and increasing Asian import competition. The 2025 closure of LyondellBasell/Covestro's Maasvlakte SMPO unit is a clear indicator of this trend.
The POC (Cumene) process and the HPPO process are the preferred technologies for all new world-scale capacity above 200,000 t/y, as both are co-product-free and benefit from simplified economics. POC holds a structural energy advantage over HPPO in high energy-cost environments, as cumene's latent heat of vaporization (330 kJ/kg) is significantly lower than methanol's (1,100 kJ/kg), reducing solvent recovery costs. HPPO holds a capital advantage and benefits from ongoing Evonik HP plant co-location as a fully integrated licensing package.
A pre-commercial in-situ H2O2 route — generating H2O2 from H2 and O2 directly in the epoxidation reactor over Pd/TS-1 — is under development by LyondellBasell and BASF, but remains limited by low per-pass productivity and catalyst deactivation challenges.
References
- Kawabata T. et al. (2019). Trends and Views in the Development of Technologies for Propylene Oxide Production. Sumitomo Kagaku.
- Tsuji J. et al. (2006). Development of New Propylene Oxide Process. Sumitomo Kagaku.
- Bassler P., Weidenbach M. and Goebbel H. (2010). The new HPPO Process for Propylene Oxide: From Joint Development to Worldscale Production. Chemical Engineering Transactions, 21, 571–576. DOI: 10.3303/CET1021096.
- Lew D. (2023). From the Dream Reaction to the Real Process: the Implemented HPPO Process. Sustainable Chemistry.
- Nexant ChemSystems PERP Program (2013). Propylene Oxide PERP 2012-7. Nexant Inc.
- HPPO Technology — Evonik Active Oxygens.
- Propylene Oxide (HPPO) — thyssenkrupp Uhde.
- Yang J. et al. (2024). Review and perspectives on TS-1 catalyzed propylene epoxidation. iScience, 27(3), 109064. DOI: 10.1016/j.isci.2024.109064.
- Alvear M. et al. (2021). Study of the Product Distribution in the Epoxidation of Propylene over TS-1 Catalyst in a Trickle-Bed Reactor. Ind. Eng. Chem. Res., 60(6), 2430–2438. DOI: 10.1021/acs.iecr.0c06150.
- Kalyoncu S. (2012). Catalytic Reaction of Propylene to Propylene Oxide on Various Catalysts. Thesis, Middle East Technical University.
- Carbonio E.A. et al. (2023). Adjusting the Chemical Reactivity of Oxygen for Propylene Epoxidation on Silver by Rational Design. ACS Catal., 13(9), 5906–5913. DOI: 10.1021/acscatal.3c00297.
- Introduction to Sinopec HPPO Process (2023). Conference presentation slides. Sinopec Shanghai Engineering Co., Ltd.