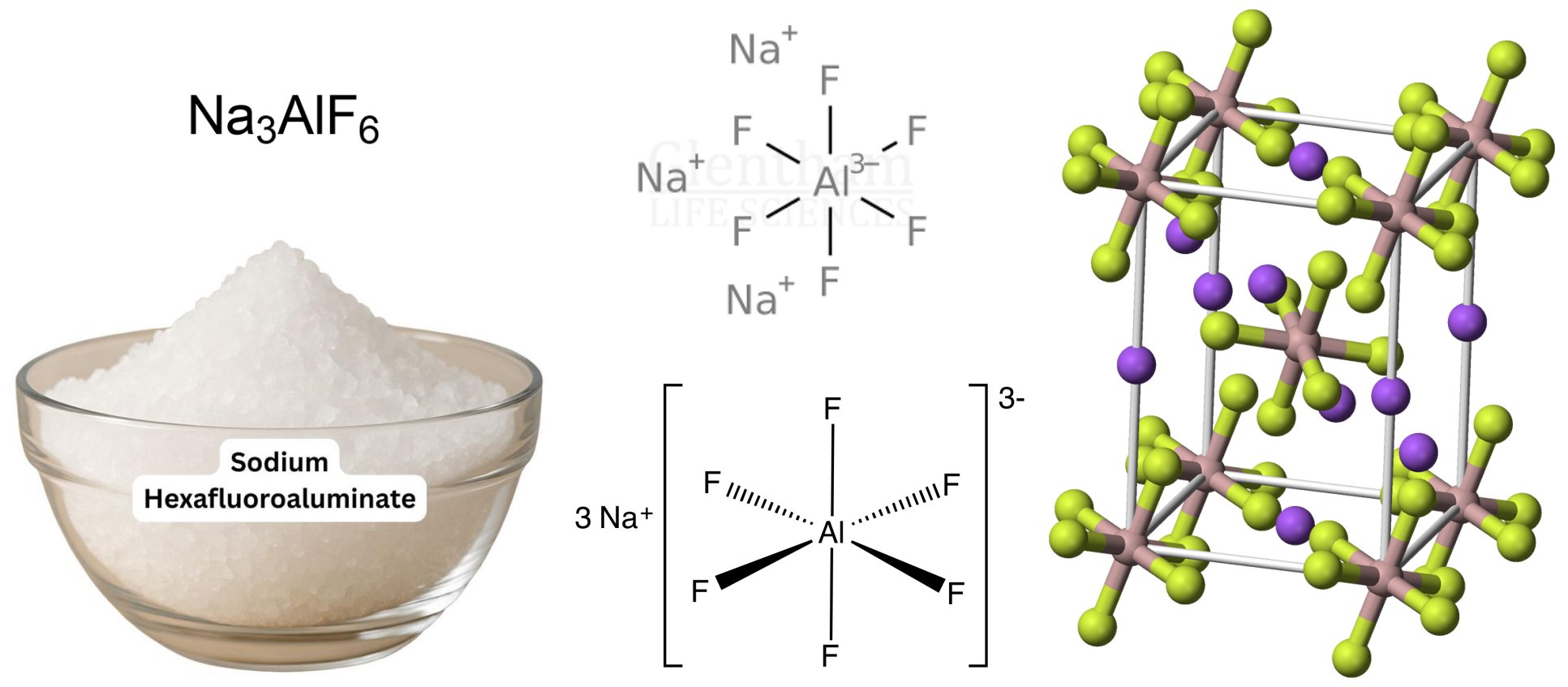
Product
- Product
- Sodium Hexafluoroaluminate (Cryolite)
- Names
- Cryolite; Trisodium Hexafluoroaluminate
-

- #PS784
- Main Product
- Aluminum Fluoride
- Segment
- Chemicals
- Main-Family
- Inorganics
- Sub-Family
- Inorganic Fluorine Compounds
- Physical State
-
Solid
Description
Your insights will be shown here
Product Communicator
| Title | Date | |
|---|---|---|

|
3/5/2026 |
Identifiers
-
 CAS Number
CAS Number
- 13775-53-6
-
 EC Number
EC Number
- 237-410-6
-
 ECHA InfoCard
ECHA InfoCard
- 100.033.995
-
 IUPAC Name
IUPAC Name
- Sodium hexafluoroaluminate
-
 PubChem ID
PubChem ID
- 159692
Chemical Data
- Chemical Formula
-
Na3AlF6
- Molecular Weight (g/mol)
- 209.94
- Melting Point (°C)
- 950
- Sulfur Content (wt%)
- 0
- Specific Gravity
- 2.90
Crude Data
- API Gravity
- -82.71
- Country
Product Settings
- Default
- Status
- A
Content provided by
| Transaction | Name | Date |
|---|---|---|
| Modified by |
|
3/5/2026 4:30 PM |
| Added by |
|
3/5/2026 3:54 PM |