Technology Overview
The Hydrisom process is a proprietary refinery pretreatment technology originally developed and licensed by Phillips Petroleum Company (Bartlesville, Oklahoma), now held by Phillips 66. It is designed as a feed-preparation unit for HF Alkylation, combining two sequential catalytic reactions in a single unit: selective hydrogenation of diolefins and olefin double-bond isomerization. The technology's name is a portmanteau of hydrogenation + isomerization, reflecting its dual function.
Technology History
During the late 1930s, Phillips Petroleum Company researchers discovered the benefits of using hydrofluoric acid to catalyze the synthesis of high-octane fuels from a broad range of low-value C3, C4, and C5 feedstocks. This led to the commercialization of the HF Alkylation process at Phillips' Borger, Texas refinery in 1942, initially to provide aviation gasoline during World War II. The Hydrisom process was subsequently developed as a complementary pretreater to improve the quality and octane potential of the mixed C4 olefin feed entering HF Alkylation units — particularly as FCC units began producing elevated butadiene concentrations in their C4 cuts, increasing acid consumption in the downstream HF unit.
By the mid-1990s, the technology gained strong international commercial traction as refineries across Latin America and beyond transitioned from leaded to unleaded gasoline production. Pemex (Mexico) licensed Hydrisom alongside four new HF Alkylation units as part of a ~$120 million modernization programme targeting 100% unleaded gasoline production, while YPF S.A. (Argentina) deployed Hydrisom at its Luján de Cuyo refinery in 1996–1997 as part of a total investment of nearly $30 million. As of 2004, ConocoPhillips had built 11 HF Alkylation units in its own refineries and had licensed over 100 grassroots units worldwide.
Technology Ownership & Licensing
The Hydrisom technology has passed through three corporate entities as a result of industry consolidation:
| Period |
Entity |
Event |
| Until 2002 |
Phillips Petroleum Company |
Original developer & licensor |
| 2002–2012 |
ConocoPhillips |
Inherited via Conoco–Phillips merger, August 2002 |
| 2012–present |
Phillips 66 |
Inherited via ConocoPhillips downstream spinoff, May 2012 |
Phillips 66 is the current owner and licensor, offering Hydrisom as a proprietary pretreater bundled within its HF Alkylation licensing package. It is important to note that Chevron Phillips Chemical (CPChem) — the 50/50 joint venture between Phillips 66 and Chevron formed in 2000 — holds only petrochemical technologies (polyethylene, aromatics, olefins) and is entirely separate from this refining technology portfolio.
Technology Summary & Chemistry
The Hydrisom unit processes a mixed C4 olefin stream — typically originating from an FCC unit — containing 1,3-butadiene, butene-1, cis- and trans-butene-2, isobutylene, n-butane, and isobutane. Without pretreatment, butadiene in the alkylation feed causes polymerization reactions in the HF acid, producing acid-soluble oil (ASO) — complex, cyclic conjunct polymers that consume HF acid, generate unwanted by-products, and reduce alkylate octane number. Two key reactions address this:
Reaction 1 — Selective Diolefin Hydrogenation:
1,3-Butadiene + H2 → Butene-1 [Catalyst: Pd/Ag/KF/Al2O3]
Butadiene (and pentadienes) are selectively hydrogenated to mono-olefins, while over-hydrogenation of butenes to butanes is suppressed by the proprietary catalyst system.
Reaction 2 — Double-Bond Isomerization:
Butene-1 → cis-Butene-2 + trans-Butene-2 [Isomerization catalyst]
Butene-1 is isomerized to the thermodynamically favored 2-butene isomers, which are the preferred HF alkylation olefins because they produce alkylate with a higher concentration of trimethylpentanes — the highest-octane C8 isomers.
The net effect is twofold: butadiene removal reduces ASO formation and lowers HF acid consumption, while the butene-1 → butene-2 shift improves alkylate Research Octane Number (RON).
Step-by-Step Process Description
The Hydrisom unit receives a mixed C4 olefin cut from the FCC unit and delivers a conditioned, diolefin-free, 2-butene-enriched C4 stream as feed to the downstream HF Alkylation unit. The process consists of two sequential catalytic steps carried out in fixed-bed reactors, followed by product stabilization.
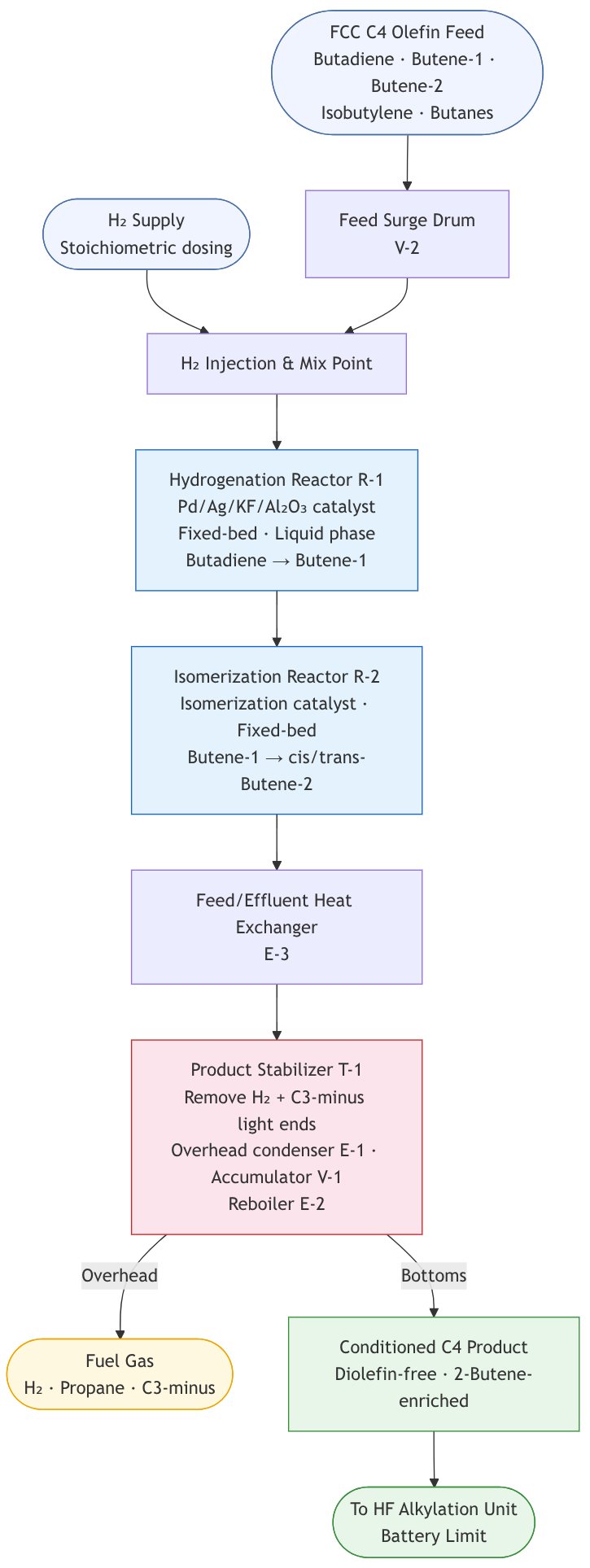
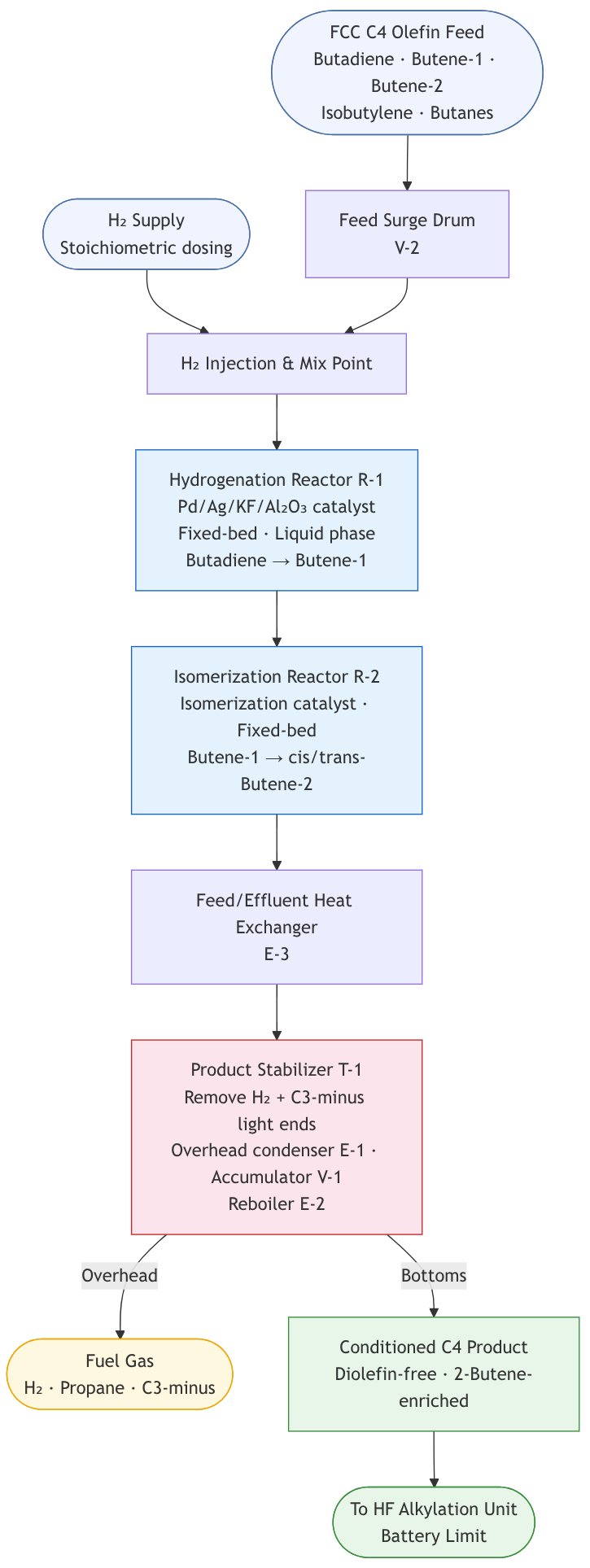
Hydrisom Block Flow Diagram (Mermaid code in the Appendix)
Step 1 — FCC C4 Olefin Feed
- Feed origin: Mixed C4 olefin cut from the FCC unit, containing 1,3-butadiene, butene-1, cis- and trans-butene-2, isobutylene, n-butane, and isobutane
- Feed contaminants of concern: 1,3-butadiene and C5 diolefins (pentadienes), whose presence in the downstream HF unit causes ASO formation — consuming HF acid, generating unwanted by-products, and reducing alkylate octane
- Typical feed composition (6,000 BPSD alkylate basis): butenes 3,068 BPSD, isobutane 2,380 BPSD, propane 115 BPSD, n-butane 702 BPSD, 1,3-butadiene 20 BPSD, pentenes 56 BPSD, propylene 153 BPSD
Step 2 — Selective Diolefin Hydrogenation (Hydrisom Reactor 1)
Based on Phillips Petroleum proprietary catalyst technology (US Patent 5,475,173A):
- Reaction: Selective hydrogenation of 1,3-butadiene (and C5 diolefins) to mono-olefins (butene-1), suppressing over-hydrogenation to paraffins
- Catalyst system:
- Palladium (Pd): 0.01–2 wt% (preferred: 0.05–0.6 wt%), as Pd metal or PdO
- Silver (Ag): 0.02–10 wt% (preferred: 0.1–5 wt%), as Ag metal or Ag oxide — selectivity modifier suppressing mono-olefin hydrogenation
- Alkali metal fluoride (preferably KF: 0.2–5 wt% K) — further selectivity modifier
- Support: alumina (Al₂O₃)
- Reactor type: Fixed-bed, liquid-phase or trickle-phase operation
- Hydrogen supply: Stoichiometric quantity of hydrogen metered precisely to react with diolefins only; excess hydrogen is minimized to protect mono-olefin yield
- Result: Butadiene and pentadienes reduced to very low levels; stream enriched in butene-1
Step 3 — Double-Bond Isomerization (Hydrisom Reactor 2)
Based on Phillips Petroleum process philosophy (US Patent 4,581,474A):
- Reaction: Double-bond (positional) isomerization of butene-1 to cis- and trans-2-butene — distinct from skeletal isomerization
- Rationale: 2-butenes produce alkylate with higher trimethylpentane concentration and higher octane than butene-1
- Result: Product stream substantially free of 1,3-butadiene and C5 diolefins, enriched in cis/trans-2-butene
Step 4 — Product Stabilizer
- The reactor effluent is sent to a stabilizer/stripper column to remove dissolved hydrogen and C3-minus light ends
- Overhead: Light ends (propane, C3-minus) and dissolved H₂ routed to fuel gas
- Bottoms: Conditioned C4 stream — diolefin-free and 2-butene-enriched — constitutes the Hydrisom unit product, transferred forward as olefin feed to the HF Alkylation unit
Equipment List
The following equipment items constitute the Hydrisom unit:
Reactors
- Hydrogenation reactor (R-1): Fixed-bed catalytic reactor; liquid-phase or trickle-phase operation; loaded with proprietary Pd/Ag/KF/Al₂O₃ catalyst for selective diolefin hydrogenation
- Isomerization reactor (R-2): Fixed-bed catalytic reactor; double-bond isomerization of butene-1 to cis/trans-2-butene
Separation Equipment
- Product stabilizer/stripper column (T-1): Distillation column to remove dissolved hydrogen and C3-minus light ends from reactor effluent; overhead routed to fuel gas, bottoms is the conditioned C4 product
- Stabilizer overhead condenser (E-1): Shell-and-tube heat exchanger for overhead vapor condensation
- Stabilizer overhead accumulator drum (V-1): Reflux drum for stabilizer overhead liquid/vapor separation
- Stabilizer reboiler (E-2): Shell-and-tube heat exchanger providing stabilizer column bottoms heat duty
Heat Exchange Equipment
- Feed/effluent heat exchanger(s) (E-3): Preheat of reactor feed against reactor effluent for energy integration
- Reactor feed cooler/trim cooler (E-4): Cooling of feed to reactor inlet temperature
Vessels & Drums
- Feed surge drum (V-2): Inlet feed surge/buffer vessel for FCC C4 olefin stream
- Hydrogen injection system / mix drum: Controlled addition and mixing of stoichiometric hydrogen into the C4 feed stream upstream of the hydrogenation reactor
Pumps & Compression
- Feed pump (P-1): Charges liquid C4 olefin feed to reactor train at required pressure
- Reflux pump (P-2): Returns stabilizer reflux
- Product rundown pump (P-3): Transfers conditioned C4 product to the downstream HF Alkylation unit battery limit
Utilities & Ancillary
- Hydrogen supply connection: Tie-in to refinery hydrogen header or dedicated hydrogen source for precise stoichiometric H₂ dosing
- Instrument air, nitrogen, utilities connections: Standard refinery utility headers
Process Efficiency
| Parameter |
Value / Description |
| Butadiene/pentadiene removal |
Reduced to very low levels (near-complete conversion) |
| Mono-olefin yield preservation |
100% — no net loss of butenes to butanes by design |
| Alkylate RON (HF alky, treated feed) |
95.6 RON (Research), 94.1 MON (Motor), clear |
| Alkylate RVP |
5.0 lb/in² Reid Vapor Pressure |
| Alkylate olefin content |
0 vol% |
| Alkylate sulfur content |
5 wt ppm |
| ASO production |
8 BPSD from 6,000 BPSD alkylate plant |
Commercial Experience
- Phillips 66: Phillips Petroleum / ConocoPhillips had built 11 HF Alkylation units across its own refineries; all of these, along with the Hydrisom and HF Alkylation technology licensing portfolio, transferred to Phillips 66 upon the May 2012 spinoff. These include the Borger, Texas Refinery, USA, site of the world's first commercial HF Alkylation unit (1942), where the foundational Hydrisom pretreatment process was subsequently developed.
- Worldwide licensed units: Over 100 grassroots HF Alkylation units licensed worldwide with Hydrisom as standard pretreater as of 2003 when worldwide alkylation capacity exceeded 1.81 million bbl/day, with HF-based processes accounting for ~57% of total at this time.
- YPF — Luján de Cuyo Refinery, Mendoza, Argentina: Hydrisom started up December 1996, HF Alkylation January 1997, both rated at 2,500 b/d; enabled elimination of leaded gasoline and export of 95 RON (Super) and 97 RON (Ultra) unleaded grades
- Pemex — Multiple Mexican Refineries: Hydrisom contracted alongside four new HF Alkylation units as part of Pemex's ~$120 million modernization programme in the mid-1990s
- United States and Spain: YPF engineers visited operating HF Alkylation/Hydrisom units in the United States and Spain during pre-startup training, confirming deployments in both countries
- SAMREF (Saudi Aramco Mobil Refinery Company Ltd.) — Yanbu Industrial City, Saudi Arabia: This 50/50 Saudi Aramco / ExxonMobil joint venture (400,000+ b/d) operates a Hydrisom unit paired with HF Alkylation as part of its FCC downstream train.
Recent Licensing Activities
While Phillips 66 remains the legal owner of Hydrisom, ReVAP, and HF Alkylation intellectual property, the company does not maintain an active public-facing technology licensing programme for these technologies as of 2026. Phillips 66's current technology licensing activities are limited to environmental and compliance-related technologies. The decline in new HF Alkylation construction globally — accelerated by the 2019 Philadelphia Energy Solutions refinery explosion and growing regulatory pressure on HF use — has substantially reduced the addressable market for new Hydrisom and ReVAP licences.
References
- Cheung, T.-T. P., & Johnson, M. M. (inventors); Phillips Petroleum Company (assignee). (1994, July 19). Hydrogenation process and catalyst therefore (U.S. Patent No. 5,475,173). U.S. Patent and Trademark Office.
- Hutson, T., Jr., & Hann, P. D. (inventors); Phillips Petroleum Company (assignee). (1985, Mar 11). Hydrocarbon conversion process (U.S. Patent No. 4,581,474). U.S. Patent and Trademark Office.
- Gravley M.L. (2004). ConocoPhillips reduced volatility alkylation process (ReVAP). In R. A. Meyers (Ed.), Handbook of petroleum refining processes (3rd ed., Chap. 1.7, pp. 1.79–1.89). McGraw-Hill.
- Basta, N. (1998, March 19). YPF uses Phillips technology to maximize unleaded gasoline production. Hydrocarbon Online.
- n.d. (1998, July 24). Pemex chooses Phillips Petroleum technology for alkylation. Hydrocarbon Online.
- n.d. (1998, March 16). Argentine refinery becomes an unleaded gasoline exporter. Oil & Gas Journal.
- Wikipedia. (Page version: 2026, January 28). Phillips Petroleum Company. Wikipedia, The Free Encyclopedia.
- Researchandideas (Page version: 2014, April 17). Creation of Phillips 66. ResearchandIdeas.com.
- Reuters. (2012, May 1). ConocoPhillips completes spinoff of refining business. Reuters.
- n.d. (2012, May 21). Phillips 66 licenses technology to Reliance for India gasification project. Hydrocarbon Processing.
- Phillips 66. (Accessed 2026, April 10). Phillips 66 corporate history. Phillips 66 Corporate Website.
- Samref. (Accessed 2026, April 10). Refinery Units. Samref website.
- Basta, N. (1998, December 8). Cenco Refining Licenses Phillip's ReVap Alkylation Technology Hydrocarbon Online.
- da Silva, M.W. (2026, Mar 25). New Naphtha Alkylation Technologies as Strategy to Balance Process Safety and Gasoline Quality Requirements -Eliminating the Hazard. Linkedin
- Phillips 66. (Accessed 2026, April 10). Energy Research & Innovation. Phillips 66 Corporate Website.
flowchart TD
A([FCC C4 Olefin Feed\nButadiene · Butene-1 · Butene-2\nIsobutylene · Butanes]) --> B[Feed Surge Drum\nV-2]
H2([H₂ Supply\nStoichiometric dosing]) --> C
B --> C[H₂ Injection & Mix Point]
C --> D[Hydrogenation Reactor R-1\nPd/Ag/KF/Al₂O₃ catalyst\nFixed-bed · Liquid phase\nButadiene → Butene-1]
D --> E[Isomerization Reactor R-2\nIsomerization catalyst · Fixed-bed\nButene-1 → cis/trans-Butene-2]
E --> F[Feed/Effluent Heat Exchanger\nE-3]
F --> G[Product Stabilizer T-1\nRemove H₂ + C3-minus light ends\nOverhead condenser E-1 · Accumulator V-1\nReboiler E-2]
G -->|Overhead| I([Fuel Gas\nH₂ · Propane · C3-minus])
G -->|Bottoms| J[Conditioned C4 Product\nDiolefin-free · 2-Butene-enriched]
J --> K([To HF Alkylation Unit\nBattery Limit])
style A fill:#f0f4ff,stroke:#4a6fa5
style H2 fill:#f0f4ff,stroke:#4a6fa5
style D fill:#e3f2fd,stroke:#1565c0
style E fill:#e3f2fd,stroke:#1565c0
style G fill:#fce4ec,stroke:#c62828
style I fill:#fff8e1,stroke:#f0a500
style J fill:#e8f5e9,stroke:#388e3c
style K fill:#e8f5e9,stroke:#388e3c