Historical Background
The Bayer process was invented and patented in 1887–1888 by Carl Josef Bayer, an Austrian chemist working in Saint Petersburg, Russia. His original objective was not aluminium production but supplying alumina as a mordant for dyeing cotton textiles. Bayer's key discovery was that aluminium hydroxide precipitating from alkaline solution formed crystalline particles that were easy to filter and wash, unlike the gelatinous precipitate obtained from acid-neutralisation methods used previously.
The predecessor technology was the Deville–Péchiney process (1859), developed by Henri Étienne Sainte-Claire Deville, which involved heating bauxite in sodium carbonate at 1,200°C and precipitating aluminium hydroxide with CO2. The Bayer process rapidly displaced it due to lower energy requirements and higher purity. The major electrolytic step — the Hall–Héroult process for smelting alumina to aluminium — was independently invented in 1886 by Charles Hall and Paul Héroult, creating the demand that made the Bayer process commercially essential.
Engineering improvements from 1967 onwards in Germany and Czechoslovakia modernised the process by introducing large autoclaves, heat exchangers and flash tanks to improve energy recovery, reducing specific energy consumption by approximately 30% over the following decades. Today, the Bayer process accounts for virtually 100% of global alumina production, with over 135 million tonnes per year produced.
Process Summary
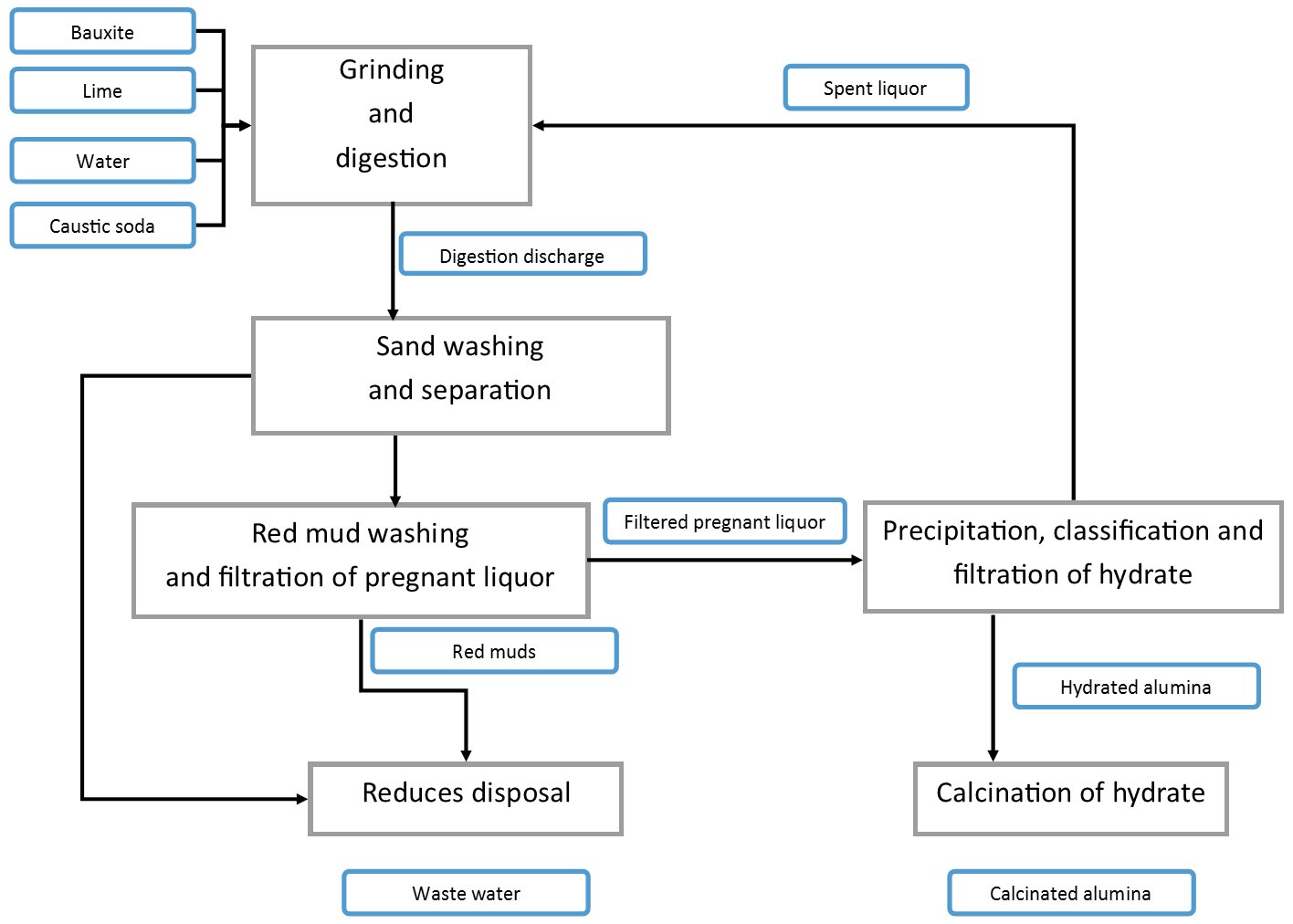
The Bayer process is a closed-loop hydrometallurgical process that selectively dissolves the aluminium oxide minerals in bauxite using hot concentrated sodium hydroxide (NaOH) solution, separates the insoluble impurities, then crystallises aluminium hydroxide from the supersaturated liquor and calcines it to produce anhydrous alumina (Al2O3). Bauxite contains 30–60% Al2O3, with the balance being iron oxides, silica, titanium dioxide and other minerals.
The process exploits the amphoteric nature of aluminium oxide — its high solubility in strongly alkaline solution at elevated temperature, contrasted with the insolubility of iron, titanium and calcium compounds under the same conditions. Two to three tonnes of bauxite are required to produce one tonne of alumina. Over 90% of alumina produced is consumed by aluminium smelters as feed for the Hall–Héroult process, with the remainder used in ceramics, abrasives, refractories, chemicals and pharmaceuticals.
Chemistry
The core reactions across each step are as follows:
- Digestion — dissolution of alumina minerals: Depending on bauxite mineralogy, one of three aluminium-bearing phases is dissolved:
Gibbsite (Al(OH)3 or AlO(OH)·H2O):
Al(OH)3 + NaOH → NaAl(OH)4
Boehmite (γ-AlO(OH)):
AlO(OH) + NaOH + H2O → NaAl(OH)4
Diaspore (α-AlO(OH)):
same stoichiometry as boehmite but requires higher temperature/pressure
General net digestion equation:
Al2O3⋅2H2O + 2 NaOH → 2 NaAlO2 + 3 H2O
- Silica side reaction — silica dissolves but is subsequently precipitated as sodium aluminium silicate (desilication product), which consumes both NaOH and alumina and reduces yield:
2 NaOH + SiO2 → Na2SiO3 + H2O
- Precipitation — crystallisation of aluminium hydroxide:
NaAl(OH)4 → Al(OH)3 + NaOH
- Calcination — dehydration to anhydrous alumina (∼1000°C):
2 Al(OH)3 → Al2O3 + 3 H2O
Process Steps, Conditions & Parameters (PFD-Based)
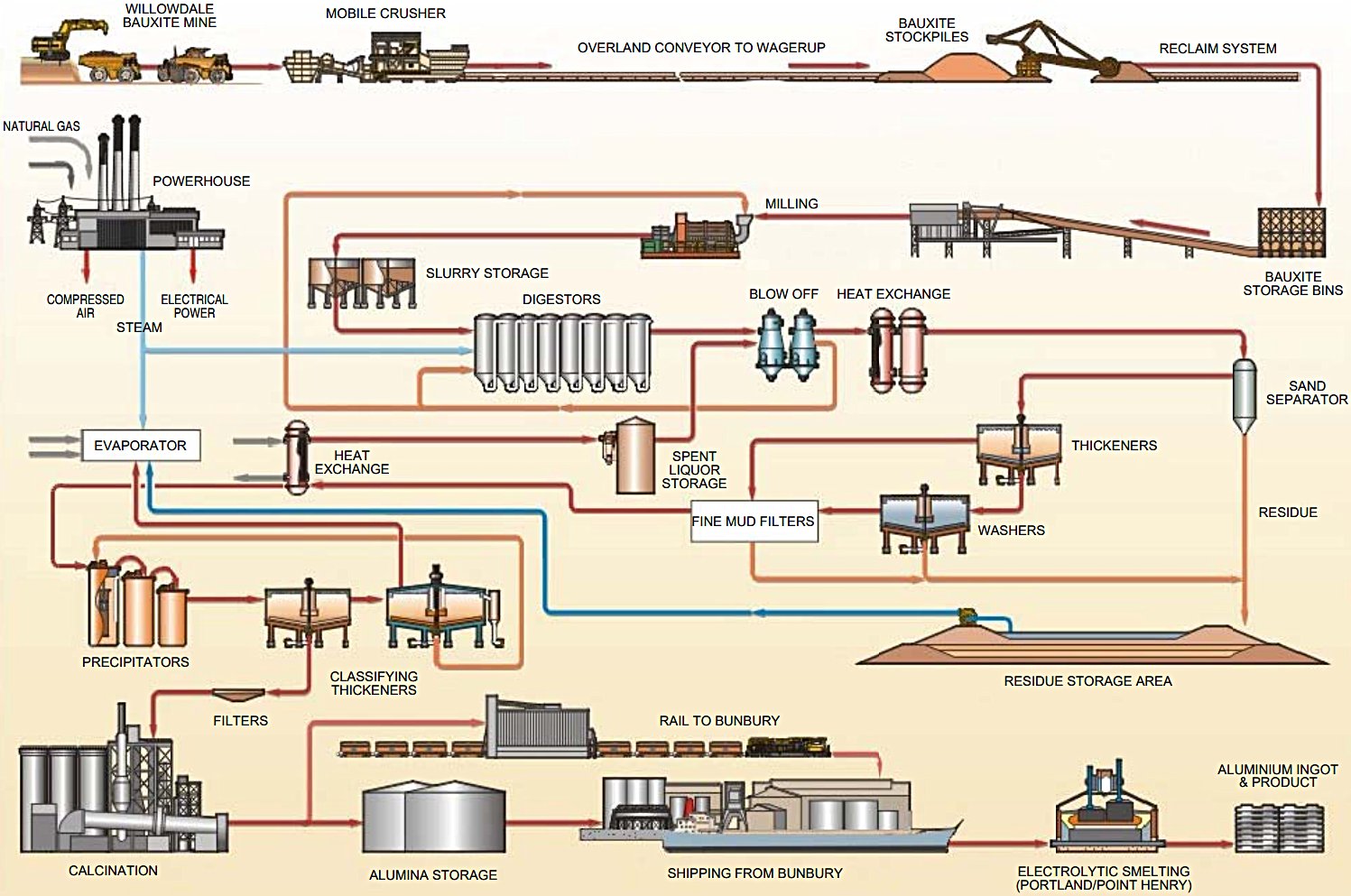
Figure 1 — Bayer Process Block Flow Diagram | Source: Environmental Protection Agency (EPA) of Ireland (Apr 2019)
The Bayer process follows a continuous loop of six principal stages:
Step 1 — Ore Preparation (Crushing & Grinding)
Bauxite is crushed at the mine and conveyed to the refinery, where it is ground in SAG (semi-autogenous) and/or ball mills to a particle size of ≤1.5 mm. Hot recycled caustic liquor is added during milling to produce a pumpable slurry (~40–55% solids). The slurry is transferred to holding/slurry storage tanks to buffer supply interruptions and begin desilication — the early precipitation of reactive silica as sodium aluminium silicate before the main digestion circuit.
| Parameter |
Value |
| Target particle size |
≤1.5 mm |
| Slurry solids concentration |
~40–55 wt% |
| NaOH addition |
From recycled spent liquor |
Step 2 — Digestion
The bauxite slurry is pumped through preheaters and into pressurised autoclave digesters, where additional concentrated NaOH is added. Temperature and pressure depend on bauxite mineralogy:
| Mineral Phase |
Temperature |
Pressure |
| Gibbsite |
135–150°C |
Near-atmospheric |
| Boehmite |
205–245°C |
Elevated (~1.5–3.5 MPa) |
| Diaspore |
>250°C |
~3.5 MPa (~35 atm) |
The alumina dissolves selectively to form sodium tetrahydroxoaluminate (green liquor/pregnant liquor), while iron oxides, titanium oxide and calcium compounds remain undissolved. Lime (CaO) may be added to precipitate silica as calcium silicate, protecting yield. Residence time in digesters is typically 30–60 minutes.
Step 3 — Clarification (Solid–Liquid Separation)
The hot slurry is cooled and transferred through flash tanks (which recover steam/heat) to large gravity settling thickeners (mud thickeners). The undissolved solids — red mud (predominantly iron oxide, silica, titania, unreacted alumina) — settle and are separated from the clarified pregnant liquor. Flocculants such as starch are added to improve settling of fine particles. The pregnant liquor is polished through security filters (leaf or pressure filters) to eliminate fine solids before precipitation.
The red mud underflow is washed in a counter-current decantation (CCD) washing train to recover caustic soda, which is recycled. Washed residue is pumped to residue disposal areas (RDA).
| Parameter |
Value |
| Thickener diameter |
Up to 60 m+ |
| Flocculant |
Starch or synthetic polymer |
| Red mud generation |
~1.0–2.0 t dry/t alumina (varies by bauxite) |
| Caustic recovery |
>95% by CCD washing |
Step 4 — Precipitation (Crystallisation)
The clarified pregnant liquor (supersaturated in NaAlO2) is cooled progressively through plate heat exchangers, transferring heat to the cold spent liquor return stream. The cooled liquor is seeded with fine Al(OH)3 crystals from previous cycles in large precipitation tanks (precipitators) operating in series. Crystals nucleate on the seeds, grow and agglomerate over a residence time of 24–72 hours. The slurry is then classified:
- Coarse fraction (product hydrate) → calcination
- Fine fraction (seed crystals) → recycled back to precipitation tank feed
- Spent liquor → evaporation and return to digestion
| Parameter |
Value |
| Precipitation temperature |
55–75°C (decreasing through chain) |
| Seed ratio |
300–600 g seed/L liquor |
| Residence time |
24–72 hours |
| Al(OH)3 recovery |
~90% of dissolved alumina |
| Precipitator tank volume |
Up to 10,000 m³ each |
Step 5 — Evaporation (Liquor Reconcentration)
Spent liquor from precipitation is dilute in NaOH and must be reconcentrated before returning to digestion. This is achieved in multi-effect evaporator trains using steam heat. Evaporation also concentrates organic impurities (primarily sodium oxalate) that build up in the circuit; these are managed by oxalate seeding and precipitation or liquor burning in a rotary kiln.
Step 6 — Calcination
Washed and filtered Al(OH)3 hydrate is fed into high-temperature calciners to drive off chemically bound water and produce anhydrous α-Al2O3:
- At 400–600°C: γ-Al2O3 forms (chemically active)
- At 1,000°C (typical SGA production): transition aluminas
- Above 1,150°C: α-Al2O3 (corundum — inert ceramic grade)
Smelter-grade alumina (SGA) is produced at approximately 1,000°C. The final product is a white crystalline powder with particle size 0.5–10 μm and Na2O content of 300–7,000 ppm.
| Parameter |
Value |
| Calcination temperature (SGA) |
950–1,050°C |
| Equipment |
Rotary kilns or CFB flash calciners |
| Calcination energy |
~1.4 GJ/t alumina (theoretical) |
| Product phase |
α-Al2O3 (SGA) |
Key Equipment & Devices
Figure 2 — Bayer Process Flow Diagram - Wagerup refinery expansion | Source: Alcoa (Aug 2003)
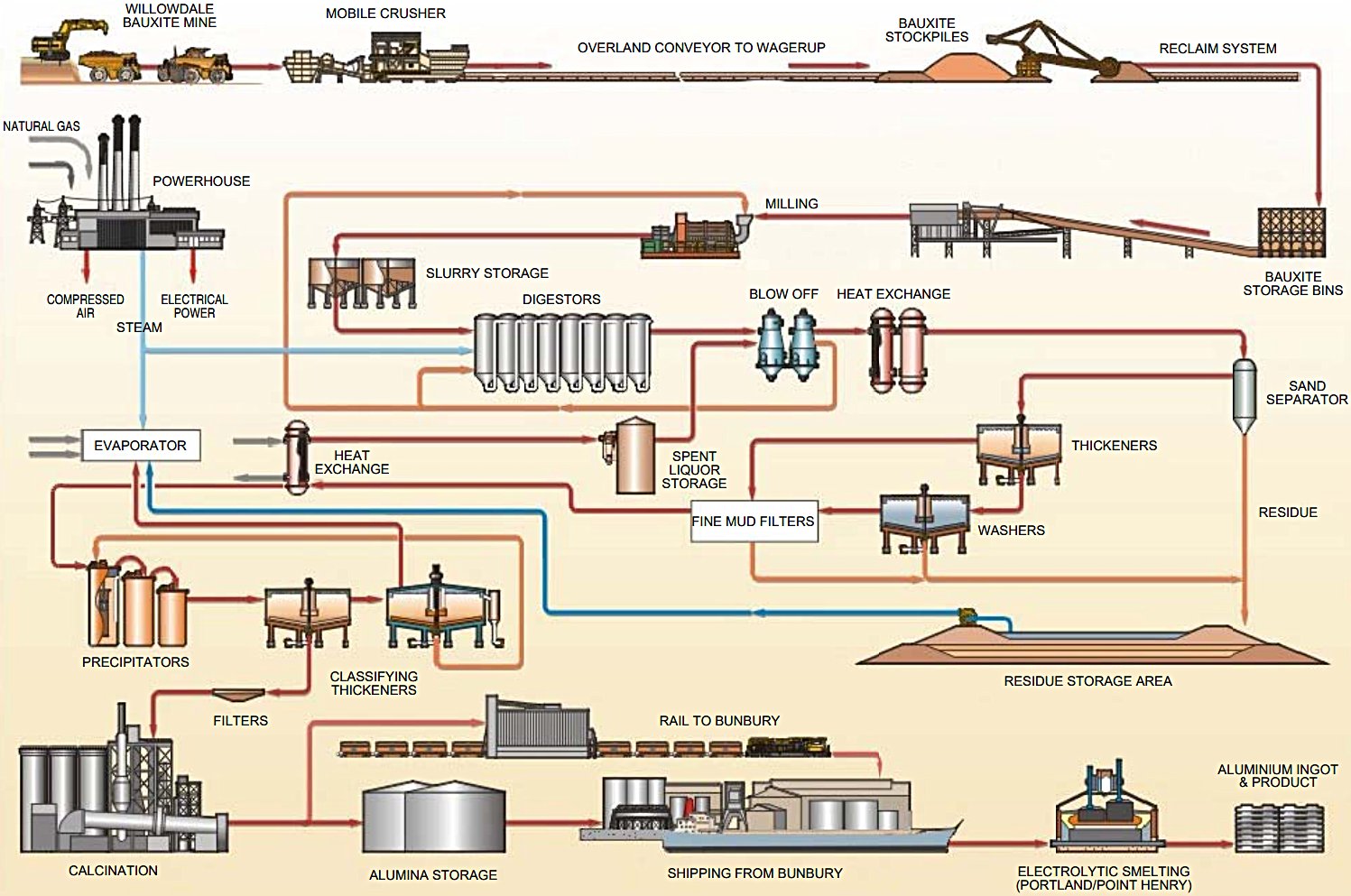
| Equipment |
Function |
Key Specifications |
| SAG/Ball Mills |
Bauxite grinding to slurry |
Particle size target ≤1.5 mm |
| Slurry Storage Tanks |
Buffer storage & pre-desilication |
Agitated; lined steel vessels |
| Preheaters (Shell & Tube HX) |
Slurry preheat before digester |
Steam heated; recovers flash steam |
| Digesters / Autoclaves |
High-T/P dissolution of alumina |
135–250°C, up to 35 atm; series-connected |
| Flash Tanks |
Pressure let-down + heat recovery |
Multi-stage; steam recovered to preheaters |
| Rotary Sand Trap |
Remove coarse undissolved solids |
Before thickeners |
| Mud Thickeners (Settlers) |
Red mud gravity settling |
Rake thickeners, diameter up to 60 m+ |
| CCD Washing Train |
Caustic recovery from red mud |
Counter-current decantation; multiple stages |
| Security/Leaf Filters |
Final polishing of pregnant liquor |
Pressure leaf or vacuum drum filters |
| Plate Heat Exchangers |
Cool pregnant liquor before precipitation |
Transfers heat to spent liquor return |
| Precipitator Tanks |
Al(OH)3 crystallisation |
Up to 10,000 m³ each; 8–20 tanks in series |
| Classifying Thickeners |
Separate seed from product hydrate |
Gravity settling; conical bottom |
| Hydrate Filters |
Dewater Al(OH)3 before calcination |
Vacuum or pressure drum filters |
| Multi-Effect Evaporators |
Reconcentrate spent caustic liquor |
Steam-driven; 3–7 effects |
| Rotary Kilns / CFB Calciners |
Calcine Al(OH)3 to Al2O3 |
950–1050°C; gas-fired |
| Electrostatic Precipitators (ESP) |
Capture alumina dust from calciners |
On each calciner stack |
| Boilers / Steam System |
Generate process steam |
High-pressure and low-pressure steam mains |
| Oxalate Kilns |
Destroy organic impurities |
Rotary kiln; oxalate → carbonate |
Process Efficiency & Economics
Material Efficiency
- Bauxite consumption: 1.7–3.3 tonnes per tonne of alumina (typically ~2–2.5 t/t for high-grade tropical bauxites)
- Alumina extraction yield: ~90% of soluble alumina in the ore is recovered
- Caustic soda (NaOH): Recycled in closed loop; make-up required only to replace losses (~40–60 kg NaOH/t alumina)
- Red mud generation: ~1.0–2.0 dry tonnes per tonne of alumina, depending on bauxite silica content
Energy Consumption
- Total specific thermal energy: approximately 10 GJ/t alumina (thermal) + 0.65 GJ/t (electrical) at a typical European refinery
- Theoretical minimum (chemistry only) is <1 GJ/t; the gap reflects heat losses in physical processes
- Calcination alone accounts for ~1.4 GJ/t alumina thermodynamically
- Best-practice refineries (e.g., Hydro's Alunorte in Brazil using high-quality gibbsitic bauxite at lower digestion temperatures) achieve significantly better energy figures
- Energy consumption has decreased by approximately 30% since the 1970s due to heat integration improvements
Economics
- Current production costs vary widely by energy source, bauxite quality and location; natural gas-fired refineries have lower costs than coal or heavy oil-fired ones
- The global alumina refining market was estimated at USD 47.5 billion in 2024, projected to reach USD 69.3 billion by 2034 at a CAGR of 3.5%
- The Bayer process holds approximately 85% of global alumina refining market revenue share in 2025
- Bauxites with >10% silica become uneconomic under the Bayer process because silica forms insoluble sodium aluminium silicate that consumes caustic and alumina, requiring alternative processes
Global Market Deployments
The Bayer process is deployed on every continent where bauxite is available. Major producing countries and flagship refineries include:
| Country / Region |
Key Refineries / Operators |
Notes |
| China |
Chalco, Weiqiao, Hongqiao, Tianli |
79.8 Mt alumina produced in 2023; world's largest producer |
| Australia |
Alcoa (Pinjarra, Wagerup, Kwinana), South32 (Worsley), Rio Tinto (QAL, Yarwun) |
Large-scale gibbsitic bauxite; major export hub |
| Brazil |
Hydro Alunorte (Barcarena) |
World's largest single alumina refinery; ~6.3 Mtpa |
| India |
NALCO (Damanjodi), Vedanta (Lanjigarh), Hindalco |
Growing capacity; government strategic investment |
| Jamaica |
Jamalco, Windalco |
Long-established Caribbean producers |
| Guinea / West Africa |
Compagnie des Bauxites de Guinée |
Mostly bauxite export; alumina refining growing |
| Ireland |
Aughinish Alumina (Rusal) |
~2 Mtpa; Europe's largest alumina refinery |
| Hungary |
Ajka (formerly) |
Notable for 2010 red mud dam breach incident |
| Turkey |
Eti Aluminium (Seydisehir) |
Operating since 1973 |
| Germany / Europe |
TRIMET, former Hamburger Aluminium-Werk |
Integrated smelter-refinery operations |
China accounted for the majority of global alumina production in 2023 with 79.8 Mt, operating close to an installed capacity of 100 Mtpa.
Technology Licensors & Key Equipment Suppliers
Unlike petrochemical processes, the Bayer process itself is not proprietary — the fundamental patents expired over a century ago and the core technology is in the public domain. However, major aluminium companies (Alcoa, Rio Tinto, Hydro, Rusal) maintain extensive proprietary know-how in process optimisation, bauxite-specific digestion conditions, precipitation seed management and calcination control. Key technology providers and equipment suppliers for major Bayer process sections are:
| Provider |
Role / Technology Supplied |
| Metso Outotec (now Metso) |
Industry leader for CFB (Circulating Fluidized Bed) calciners — described as the industry standard; also supplies hydrate filtration, flash evaporation and digestion equipment |
| Alcoa |
Proprietary Bayer variants (A-2 and other processes) for specific bauxite types; extensive internal R&D on precipitation and digestion optimisation ? |
| Norsk Hydro |
Hydro Alunorte process optimisation; energy-efficient digestion for gibbsitic bauxite ? |
| Rio Tinto / Alcan |
AP-Technology integration; process know-how for Yarwun and QAL refineries |
| Rusal |
Proprietary modifications for diaspore/boehmite bauxites from Russian deposits (Urals region) |
| Hatch |
Engineering, project management and process design for greenfield and brownfield alumina refineries |
| Worley (WorleyParsons) |
EPCM for alumina refineries globally |
| SysCAD |
Leading process simulation / digital twin platform for Bayer process modelling and optimisation |
| IHI Corporation (Japan) |
Fluid flash calciners and associated alumina refinery equipment for Asian markets |
Environmental Considerations
Red mud (bauxite residue) is the principal waste stream, generated at ~1–2 dry t per tonne of alumina, with a highly alkaline pH (~10–13) and complex chemistry including iron, aluminium, calcium, sodium and trace heavy metals. Disposal in large lined impoundments is standard in the US and many other countries, though dam failures (notably the 2010 Ajka disaster in Hungary, releasing 700,000 m³ of slurry and causing 10 deaths) highlight the environmental risk. Research into red mud valorisation — ceramics, iron recovery, soil amendment — is active but commercial-scale utilisation remains limited.
Energy decarbonisation is the primary sustainability challenge, with current efforts focused on process electrification, solar thermal integration, green hydrogen combustion and low-grade heat recovery to eliminate dependence on fossil fuel-fired steam boilers. Best-practice refineries have already reduced specific CO2 emissions by ~30% versus 1970s baselines, with further reductions targeted through deeper heat integration and electrification of calciners.
References
References are temporarily parked here.