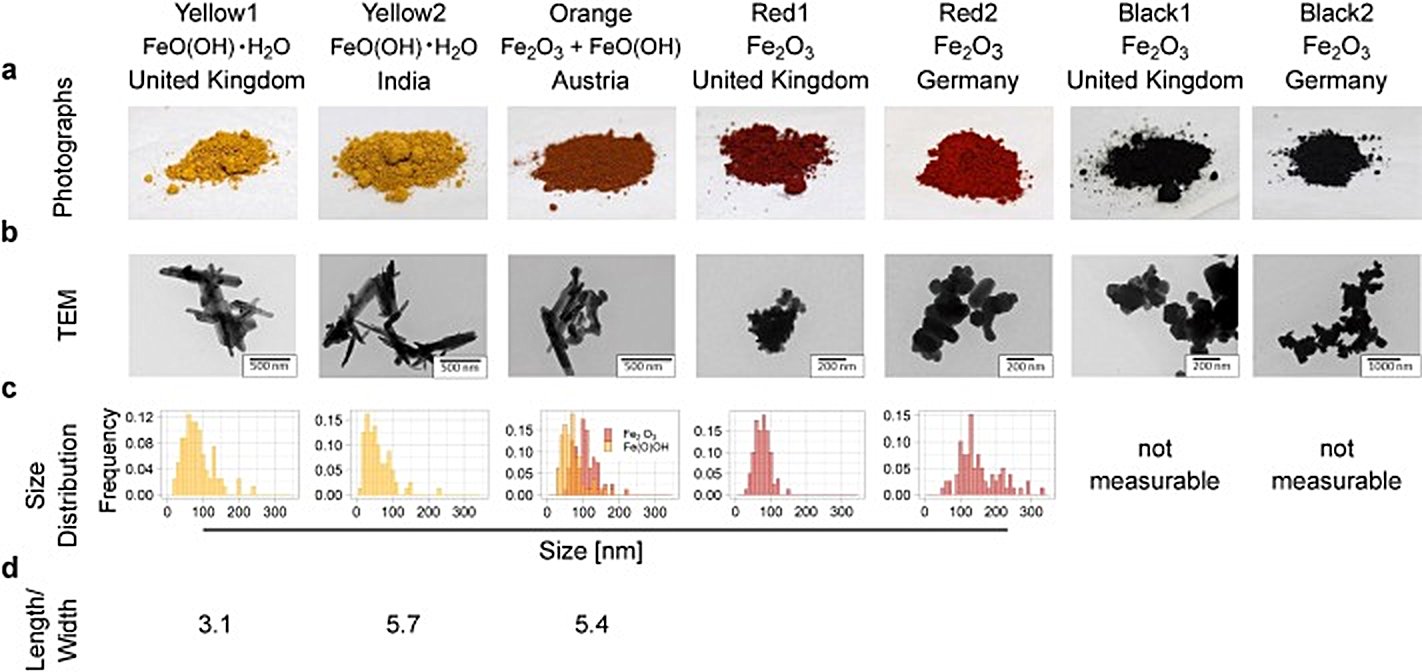
Iron oxides are a class of inorganic chemical compounds composed of iron (Fe) and oxygen (O) atoms, often also including related oxyhydroxide forms that incorporate hydrogen. There are approximately 16 known iron oxides and oxyhydroxides, making them a structurally diverse group of compounds. Iron oxides are among the most abundant and widespread inorganic compounds on Earth, playing important roles in geology, biology, and industry. They occur naturally as minerals, form as corrosion products (most famously rust), and are found in biological systems such as hemoglobin.
Chemical Structure
Iron oxides feature iron in either its ferrous (Fe²⁺) or ferric (Fe³⁺) oxidation states, or a combination of both. Structurally, they consist of close-packed arrays of oxygen anions (O²⁻) — typically in hexagonal or cubic close-packed arrangements — with iron cations occupying octahedral or tetrahedral interstitial sites. Many iron oxides are non-stoichiometric, meaning their elemental ratios do not conform to simple whole-number proportions.
Principal Forms
Iron oxides are chemical compounds, defined by their formula, crystal phase, and oxidation state. Beyond pure oxides, a related family of iron oxyhydroxides (FeOOH) incorporates hydroxide groups and is closely associated with weathering and corrosion processes.
| Primary Name |
Formula |
Mineral Synonym |
Color |
Natural Occurrence |
Iron(III) oxide
(α-phase) |
α-Fe₂O₃ |
Hematite |
Reddish-brown |
Yes — primary iron ore |
| Iron(II,III) oxide |
Fe₃O₄ |
Magnetite |
Black |
Yes — primary iron ore |
Iron(III) oxyhydroxide
(α-phase) |
α-FeOOH |
Goethite |
Yellow-
brown |
Yes —widespread; minor ore |
Iron(III) oxide
(γ-phase) |
γ-Fe₂O₃ |
Maghemite |
Reddish-brown |
Rarely, as
alteration product |
| Iron(II) oxide |
FeO |
Wüstite |
Black |
Extremely rare
at surface |
Iron(III) oxyhydroxide
(γ-phase) |
γ-FeOOH |
Lepidocrocite |
Orange |
Yes — common corrosion mineral |
Key Applications
Iron oxides' stability, low cost, and range of properties make them commercially significant across many sectors. The table below lists the principal applications by compound, ranked by breadth of industrial relevance:
| Rank |
Compound |
Key Applications |
| 1 |
α-Fe₂O₃ |
Pigments (paints, coatings, colored concrete); iron ore / steelmaking; heterogeneous catalysis; gas sensors; food colorant E172 |
| 2 |
Fe₃O₄ |
Steelmaking; magnetic storage and sensors; water treatment; heterogeneous catalysis; biomedical applications |
| 3 |
α-FeOOH |
Dominant yellow pigment (PY42) for construction and coatings; precursor to hematite and maghemite |
| 4 |
γ-Fe₂O₃ |
Magnetic recording media; ferrofluids; biomedical applications (MRI contrast agents, hyperthermia) |
| 5 |
FeO |
Metallurgical intermediate in iron ore reduction; precursor in iron and steel production |
| 6 |
γ-FeOOH |
Corrosion product; minor orange pigment; precursor in oxyhydroxide chemistry |
Beyond these compound-specific roles, iron oxides as a class are widely used in thermite reactions — where Fe₂O₃ reacts exothermically with aluminium to produce molten iron for welding and incendiary applications — and as a group contribute broadly to pigment production, generating earthy yellow, orange, red, brown, and black tones across paints, coatings, plastics, and ceramics.
References
- Ali A, Zafar H, Zia M, ul Haq I, Phull AR, Ali JS, Hussain A. Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnol Sci Appl. 2016;9:49-67. DOI: 10.2147/NSA.S99986
- Wikipedia. Iron oxide (Page version: Nov 13, 2025)
- ScienceDirect. Iron Oxide (Accessed Feb 27, 2026)
- Vedantu. Iron Oxide – Definition, Types, Formula, and Uses (Accessed Feb 27, 2026)
- Zadeh J., Discovery Alert (Apr 3, 2025). Hematite vs Magnetite: Understanding Types of Iron Ore in 2025
- Fortune Business Insights (Feb 9, 2026). Report FBI108698 — Iron Ore Market Size, Share & Industry Analysis, By Type (Hematite, Magnetite, and Others), By Application (Steel Production and Others), and Regional Forecast, 2026-2034
- Reports and Data (Dec 11, 2025). Magnetite Iron Ore Market
- JXSC (Jun 12, 2024). Mastering Hematite vs Magnetite: Expert Guide
- O'Hanlon G., Natural Pigments (Apr 15, 2018). Names for Iron Oxide Earth Pigments
- Ali, A., Zafar, H., Zia, M., ul Haq, I., Phull, A. R., Ali, J. S., & Hussain, A. (Aug 19, 2016). Synthesis, characterization, applications, and challenges of iron oxide nanoparticles. Nanotechnology, Science and Applications, 9, 49–67. DOI: 10.2147/NSA.S99986
- David, 911Metallurgist (Jan 4, 2021). Iron Oxide Pigments
- ChemAnalyst (Jan 2026). Iron Oxide Market Analysis.
- Mordor Intelligence (Mar 12, 2020). Iron Oxide Pigments Market Size & Share Analysis - Growth Trends and Forecast (2026 - 2031)